p53 in the immune response: the ace up the sleeve
The tumor suppressor p53 is a sequence-specific transcription factor that is activated in response to various cellular stresses such as DNA damage, oncogene over-expression and associated uncontrolled cell proliferation. p53 functions mainly as a transcription factor and is a key component in preventing cancer development through regulation of apoptosis, cell cycle and senescence genes thereby helping to maintain genome stability within an organism. Alterations of p53 function through mutation or misregulation in the p53 network are common features in human cancers with over 80-90% of tumors having an altered p53 pathway [
In this review we explore the interactions between p53 and the immune system. The focus is mainly on the role that wild type (WT) p53 plays in immune-related processes such as inflammation, innate and adaptive responses as well as functional interactions of p53 with NF-kB, which is considered a key regulator in immune responses. Emphasis is placed on p53 as a transcription factor in modulating expression of target genes involved in immunity pathways (see Figure 1).
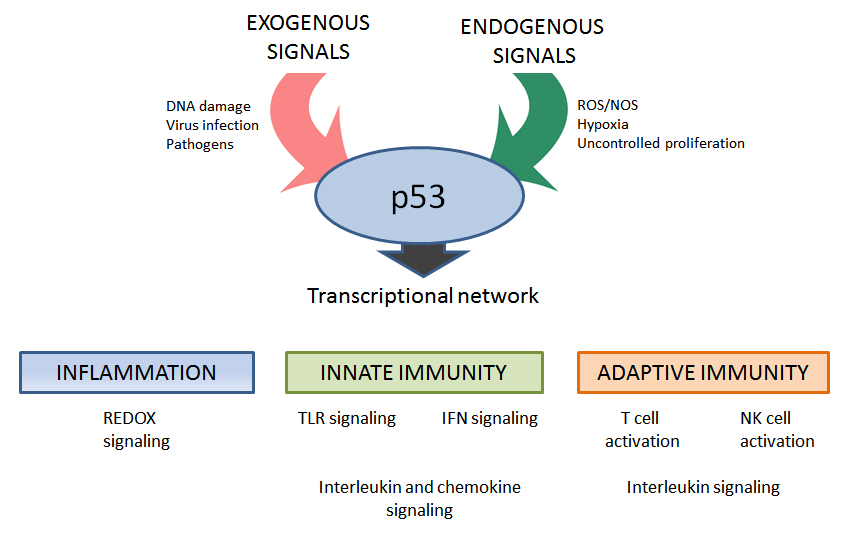
The immune system is a collection of biological processes whose tasks in preventing disease include identification and destruction of pathogens and tumor cells. Given the broad diversity in p53 controls and functions, it is not surprising that p53 touches multiple aspects of immunity. For example, DNA damage can trigger p53 responses that help orchestrate clearance of damaged cells via the innate immune system [
p53 also appears to be involved during the generation of immune cells. In agreement with its role as a modulator in stem cell appearance, p53 can limit expansion of hematopoietic stem cells (HSC) [
Inflammation and p53: maintaining homeostasis
Inflammation, a common immune response, is a protective first-responder attempt to remove injurious stimuli and to initiate healing. It is a complex signal-mediated reaction by vascular tissues to cellular insults such as pathogens and infectious agents, toxins, physical stress or damaged cells. Acute inflammation is an important mode of immune response, while chronic inflammation can cause tissue destruction or even autoimmunity.
p53 has several roles in inflammation including modifying cell growth and cellular behavior in response to DNA and inflammatory stressors. p53 is activated by DNA damage that is induced by both ROS and reactive nitrogen species (NOS) that are produced during inflammation [
As a transcription factor, p53 can modulate expression of several genes encoding enzymes involved in both production or elimination of reactive species contributing to inflammation including, for example, up-regulation of the antioxidant glutathione peroxidase (GPX1)[
Thus, p53 can play a significant role in modulating intracellular ROS/ NOS levels to aid in appropriate balance of the inflammatory responses. In addition, since p53 is subject to modifications in the presence of reactive compounds, it can be considered a cellular sensor of redox changes [
The relationship between p53 and pathogenesis of inflammation-associated cancer and other immune related diseases extends beyond induction/restriction of inflammatory responses, all of which can be affected by p53 expression, mutation or alterations in its regulatory pathway. For example, there is greater invasion of inflammatory and fibroblast cells into IR damaged tissues in p53-null compared to WT mice [
Innate and adaptive immune responses in p53 null mice can be skewed toward pro-inflammation, suggesting p53 may act as a negative regulator of inflammation (34-40). Additionally, p53 null mice are susceptible to autoimmune diseases including collagen-induced arthritis [
p53, viral infections and immune responses
Up-regulation of p53 in response to viral infections is a part of host cell defenses. For example, increased p53-dependent apoptosis can reduce viral replication [
Since the discovery over 30 years ago of p53 as a binding partner of SV40 LTag [
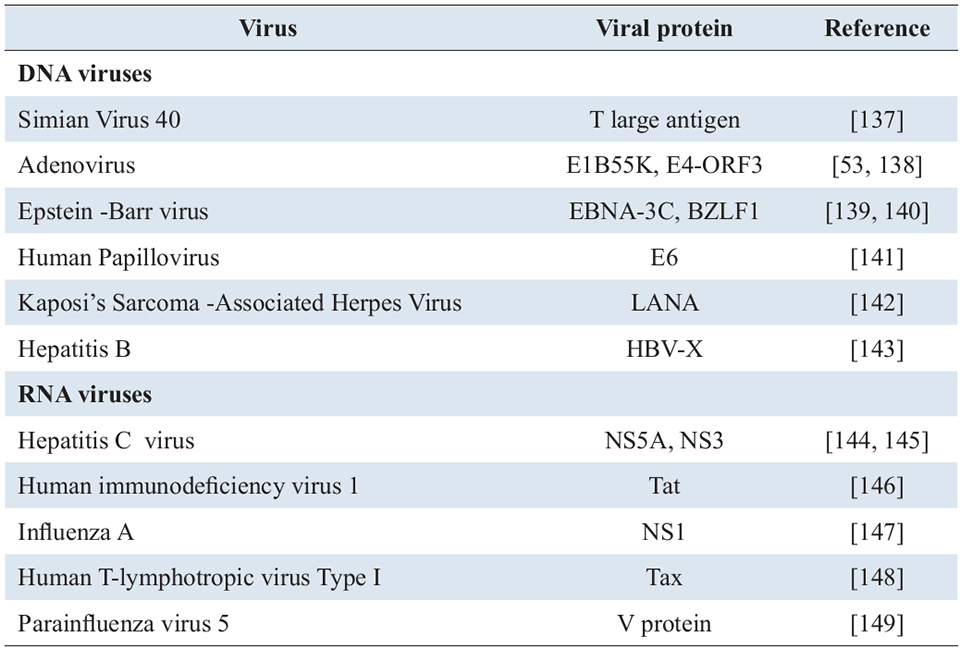
The ability of viruses to alter p53 functions and pathways is an important step in their establishment and pathogenesis in animal hosts. Described in Table 1 are examples of viral proteins that interact with p53. Viruses can disrupt p53 functions either directly or through cellular factors involved in downstream activities so as to override cell-cycle checkpoints or protect cells from p53-dependent apoptosis. p53 can be sequestered and/or inactivated by posttranslational modifications (phosphorylation, ubiquitination) induced by viral proteins or by modulation of host enzymes such as Mdm2 that promote proteasome degradation of p53 (see review by Lazo and Santos [
In response to viral infections, one of the most efficient and rapid responses triggered by the immune system is induction of type I interferon mediated signaling. This response involves activation of the STAT (signal transducer and activator of transcription) signaling pathway and subsequent expression of antiviral genes. Several years ago, the seminal discovery of Takaoka et al. [
Alternatively, p53 can influence both IFN production and signaling, enhancing the antiviral response through direct transcriptional up-regulation of several IFN-inducible genes. Included are transcriptional activators such as interferon regulatory factor 9 (IRF9) [
Overall, these findings have established important roles for p53 transcriptional activities in host defense against viral infection and support the relevance of p53 in antiviral innate immunity.
p53 general influence on immune response pathways
While there is substantial evidence that p53 protects against inflammation (mostly under chronic conditions), recent studies in mouse and human cells reveal that p53 may promote acute inflammation and immune responses ([
p53 can influence several innate and adaptive immune pathways through regulation of genes involved in signaling (chemokines, interleukins), pathogen recognition (TLRs) and activation of specific subsets of immune cells such as T and B lymphocytes, NK cells and macrophages. Interleukins and chemokines are signaling molecules that affect a variety of cellular functions and are stimulated when tissue homeostasis is altered. Both are mediators of inflammation and play critical roles in host defense by attracting and activating specific subsets of effector leukocytes, cells from the monocyte/macrophage lineage as well as natural killer (NK) cells.
Expression of chemokines and cytokines are subject to p53, depending on stimulus and cell type. p53 can increase transcription of several cytokines involved in innate immunity including colony-stimulating factor 1 (CSF1) and monocyte chemotactic protein (MCP1), chemokine CXC motif ligand (CXCL1) and interleukin 15 (IL-15) that attract macrophages, neutrophils, and natural killer cells, contributing to immune elimination of senescent cells [
Chemokines also can influence p53 activities. For example, the macrophage migration inhibitory factor (MIF), a product of activated macrophages, sustains macrophage survival and pro-inflammatory function by inhibiting p53 [
The expression of several surface markers on cells involved in immune responses is subject to p53 regulation. Genotoxic activation of p53 leads to up-regulation of intracellular-adhesion molecule-1 (ICAM-1) mRNA and protein [
Boosting innate and adaptive immune responses with p53
Recently, employing a genome-wide in silico search we found that most members of the human Toll Like Receptor (TLR) gene family contain potential p53 targets [
Using primary lymphocytes and alveolar macrophages from healthy subjects [
p53 also has transcriptional targets in antigen cell-signaling pathways of T and B lymphocytes. The TAP1 protein (transporter associated with antigen processing) is required for the major histocompatibility complex (MHC) class I antigen presentation pathway that plays a key role in host tumor surveillance. In response to DNA damage, TAP1 expression is induced by p53 in cooperation with its family member protein p73. This up-regulation enhances transport of MHC class I peptides, expression of surface MHC-peptide complexes and activation of the MHC class I pathway [
Regulation of the NK cells activities provides another example of p53 influence on the host immune system. These cells are specialized immune cells that eliminate foreign, stressed, transformed and senescent cells through specialized surface receptors, such as NKG2D [
Innate and adaptive immunity are also connected to p53 through transcriptional regulation of microRNAs (miRNAs), which are small non-coding endogenous RNAs that bind complementary sequences of target mRNAs and regulate translation of specific genes. miRNAs affect inflammation and cancer [
p53 as a direct target in cancer immunotherapy
Since the immune system must distinguish between self and non-self antigens, p53 has been considered a target for immunotherapy. First identified in the sera of cancer patients more than 30 years ago [
p53 and NF-kB cross-talk in immune responses
As described above, roles for p53 in immunity are continually emerging. However, these must be considered in light of the other well-established modulators of immunity and inflammation especially NF-kB, a master regulator of immune responses.
Most discussions of p53 and NF-kB interactions have focused on their roles in cancer. While p53 and NF-kB are generally considered to be opposing factors where p53 promotes apoptosis while NF-kB enhances survival (reviewed in [
The complicated relationship between p53 and NF-kB is also seen in the context of immune responses. p53 can play an inhibitory role in NF-kB signaling and consequently the inflammatory response. For example, p53 inhibits IKK β and NF-kB mediated transactivation in IgE-mediated degranulation of mast cells and anaphylaxis [
Positive p53/NF-kB relationships in the immune response have also been described. p53 stabilization by treatment of cells with Nutlin-3 was able to enhance retrovirus-induced apoptosis of host cells in part through augmented activation of NF-kB [
p53 and NF-kB in senescence
In recent years, both p53 and NF-kB have been shown to play a role in senescence (irreversible cell cycle arrest) based on a strong link with inflammation [

The connection between p53 and immunity: concluding remarks
p53 has an important role in innate and adaptive immune responses where activation of p53 can be both beneficial and detrimental. In addition to cancer, there are many infectious disease implications, as we had proposed for a loop between pathogen detection by TLRs, inflammation and p53 induction [
The p53/immune interaction is especially relevant to cancer as indicated in a recent review of “hallmarks of cancer” by Hanahan and Weinberg [
Although there are nearly 30 immune-related genes (including miRNAs) targeted by p53, many new targets are expected to be identified in the near future through genome-wide methods. The combination of chromatin immunoprecipitation (ChIP) with high throughput sequencing (ChIP-seq) and expression analysis has already been used to map sites of p53 binding among the hundreds of thousands of potential target sequences in the human genome and to identify candidate p53 target genes [
Acknowledgements
We thank Drs. Mike B. Fessler and Stavros Garantziotis for providing insightful comments and suggestions on the manuscript. This work was supported by the National Institute of Environmental Health Sciences, National Institutes of Health Intramural Research Program (Z01-ES065079 to MAR).
References
- Petitjean A, Mathe E, Kato S, Ishioka C, Tavtigian SV, Hainaut P et al. Impact of mutant p53 functional properties on TP53 mutation patterns and tumor phenotype: lessons from recent developments in the IARC TP53 database. Hum Mutat 2007; 28: 622-629.
Reference Link - Soussi T. The history of p53. A perfect example of the drawbacks of scientific paradigms. EMBO Rep 2010; 11: 822-826.
Reference Link - Lane D, Levine A. p53 Research: the past thirty years and the next thirty years. Cold Spring Harb Perspect Biol 2010; 2: a000893.
Reference Link - Menendez D, Inga A, Resnick MA. Potentiating the p53 network. Discov Med 2010; 10: 94-100.
- Vousden KH, Prives C. Blinded by the Light: The Growing Complexity of p53. Cell 2009; 137: 413-431.
Reference Link - Martins CP, Brown-Swigart L, Evan GI. Modeling the therapeutic efficacy of p53 restoration in tumors. Cell 2006; 127: 1323-1334.
Reference Link - Ventura A, Kirsch DG, McLaughlin ME, Tuveson DA, Grimm J, Lintault L et al. Restoration of p53 function leads to tumour regression in vivo. Nature 2007; 445: 661-665.
Reference Link - Hofseth LJ, Saito S, Hussain SP, Espey MG, Miranda KM, Araki Y et al. Nitric oxide-induced cellular stress and p53 activation in chronic inflammation. Proc Natl Acad Sci U S A 2003; 100: 143-148.
Reference Link - Moon C, Kim S, Wie M, Kim H, Cheong J, Park J et al. Increased expression of p53 and Bax in the spinal cords of rats with experimental autoimmune encephalomyelitis. Neurosci Lett 2000; 289: 41-44.
Reference Link - Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V et al. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 2007; 445: 656-660.
Reference Link - Akala OO, Park IK, Qian D, Pihalja M, Becker MW, Clarke MF. Long-term haematopoietic reconstitution by Trp53-/-p16Ink4a-/-p19Arf-/- multipotent progenitors. Nature 2008; 453: 228-232.
Reference Link - TeKippe M, Harrison DE, Chen J. Expansion of hematopoietic stem cell phenotype and activity in Trp53-null mice. Exp Hematol 2003; 31: 521-527.
Reference Link - Fuhrman LE, Goel AK, Smith J, Shianna KV, Aballay A. Nucleolar Proteins Suppress Caenorhabditis elegans Innate Immunity by Inhibiting p53/CEP-1. Plos Genetics 2009; 5: e1000657.
Reference Link - Hafsi H, Hainaut P. Redox control and interplay between p53 isoforms: roles in the regulation of basal p53 levels, cell fate and senescence. Antioxid Redox Signal 201; 15:1655-1667.
- Campbell HG, Slatter TL, Jeffs A, Mehta R, Rubio C, Baird M et al. Does Delta133p53 isoform trigger inflammation and autoimmunity? Cell Cycle 2012; 11: 446-450.
Reference Link - Donato NJ, Perez M. Tumor necrosis factor-induced apoptosis stimulates p53 accumulation and p21WAF1 proteolysis in ME-180 cells. J Biol Chem 1998; 273: 5067-5072.
Reference Link - Tan M, Li S, Swaroop M, Guan K, Oberley LW, Sun Y. Transcriptional activation of the human glutathione peroxidase promoter by p53. J Biol Chem 1999; 274: 12061-12066.
Reference Link - Yoon KA, Nakamura Y, Arakawa H. Identification of ALDH4 as a p53-inducible gene and its protective role in cellular stresses. J Hum Genet 2004; 49: 134-140.
Reference Link - de Moraes E, Dar NA, de Moura Gallo CV, Hainaut P. Cross-talks between cyclooxygenase-2 and tumor suppressor protein p53: Balancing life and death during inflammatory stress and carcinogenesis. Int J Cancer 2007; 121: 929-937.
Reference Link - Rivera A, Maxwell SA. The p53-induced gene-6 (proline oxidase) mediates apoptosis through a calcineurin-dependent pathway. J Biol Chem 2005; 280: 29346-29354.
Reference Link - Italiano D, Lena AM, Melino G, Candi E. Identification of NCF2/p67phox as a novel p53 target gene. Cell Cycle 2012; 11: 4589-4596.
Reference Link - Forrester K, Ambs S, Lupold SE, Kapust RB, Spillare EA, Weinberg WC et al. Nitric oxide-induced p53 accumulation and regulation of inducible nitric oxide synthase expression by wild-type p53. Proc Natl Acad Sci U S A 1996; 93: 2442-2447.
Reference Link - Velu CS, Niture SK, Doneanu CE, Pattabiraman N, Srivenugopal KS. Human p53 is inhibited by glutathionylation of cysteines present in the proximal DNA-binding domain during oxidative stress. Biochemistry 2007; 46: 7765-7780.
Reference Link - Maillet A, Pervaiz S. Redox regulation of p53, redox effectors regulated by p53: a subtle balance. Antioxid Redox Signal 2012; 16: 1285-1294.
Reference Link - Komarova EA, Kondratov RV, Wang K, Christov K, Golovkina TV, Goldblum JR et al. Dual effect of p53 on radiation sensitivity in vivo: p53 promotes hematopoietic injury, but protects from gastro-intestinal syndrome in mice. Oncogene 2004; 23: 3265-3271.
Reference Link - Herkel J, Mimran A, Erez N, Kam N, Lohse AW, Marker-Hermann E et al. Autoimmunity to the p53 protein is a feature of systemic lupus erythematosus (SLE) related to anti-DNA antibodies. J Autoimmun 2001; 17: 63-69.
Reference Link - Kuhn HM, Kromminga A, Flammann HT, Frey M, Layer P, Arndt R. p53 autoantibodies in patients with autoimmune diseases: a quantitative approach. Autoimmunity 1999; 31: 229-235.
Reference Link - Sun Y, Cheung HS. p53, proto-oncogene and rheumatoid arthritis. Semin Arthritis Rheum 2002; 31: 299-310.
Reference Link - Tak PP, Zvaifler NJ, Green DR, Firestein GS. Rheumatoid arthritis and p53: how oxidative stress might alter the course of inflammatory diseases. Immunol Today 2000; 21: 78-82.
Reference Link - Vousden KH, Lane DP. p53 in health and disease. Nat Rev Mol Cell Biol 2007; 8: 275-283.
Reference Link - Yamanishi Y, Boyle DL, Rosengren S, Green DR, Zvaifler NJ, Firestein GS. Regional analysis of p53 mutations in rheumatoid arthritis synovium. Proc Natl Acad Sci U S A 2002; 99: 10025-10030.
Reference Link - Hussain SP, Amstad P, Raja K, Ambs S, Nagashima M, Bennett WP et al. Increased p53 mutation load in noncancerous colon tissue from ulcerative colitis: a cancer-prone chronic inflammatory disease. Cancer Res 2000; 60: 3333-3337.
- Tapinos NI, Polihronis M, Moutsopoulos HM. Lymphoma development in Sjogren's syndrome: novel p53 mutations. Arthritis Rheum 1999; 42: 1466-1472.
Reference Link - Okuda Y, Okuda M, Bernard CC. Regulatory role of p53 in experimental autoimmune encephalomyelitis. J Neuroimmunol 2003; 135: 29-37.
Reference Link - Pesch J, Brehm U, Staib C, Grummt F. Repression of interleukin-2 and interleukin-4 promoters by tumor suppressor protein p53. J Interferon Cytokine Res 1996; 16: 595-600.
Reference Link - Santhanam U, Ray A, Sehgal PB. Repression of the interleukin 6 gene promoter by p53 and the retinoblastoma susceptibility gene product. Proc Natl Acad Sci U S A 1991; 88: 7605-7609.
Reference Link - Zheng SJ, Lamhamedi-Cherradi SE, Wang P, Xu L, Chen YH. Tumor suppressor p53 inhibits autoimmune inflammation and macrophage function. Diabetes 2005; 54: 1423-1428.
Reference Link - Komarova EA, Krivokrysenko V, Wang KH, Neznanov N, Chernov MV, Komarov PG et al. p53 is a suppressor of inflammatory response in mice. Faseb Journal 2005; 19: 1030-1032.
- Liu G, Park YJ, Tsuruta Y, Lorne E, Abraham E. p53 Attenuates lipopolysaccharide-induced NF-kappaB activation and acute lung injury. J Immunol 2009; 182: 5063-5071.
Reference Link - Takaoka A, Hayakawa S, Yanai H, Stoiber D, Negishi H, Kikuchi H et al. Integration of interferon-alpha/beta signalling to p53 responses in tumour suppression and antiviral defence. Nature 2003; 424: 516-523.
Reference Link - Munoz-Fontela C, Garcia MA, Garcia-Cao I, Collado M, Arroyo J, Esteban M et al. Resistance to viral infection of super p53 mice. Oncogene 2005; 24: 3059-3062.
Reference Link - Lane DP, Crawford LV. T antigen is bound to a host protein in SV40-transformed cells. Nature 1979; 278: 261-263.
Reference Link - DeLeo AB, Jay G, Appella E, Dubois GC, Law LW, Old LJ. Detection of a transformation-related antigen in chemically induced sarcomas and other transformed cells of the mouse. Proc Natl Acad Sci U S A 1979; 76: 2420-2424.
Reference Link - Linzer DI, Levine AJ. Characterization of a 54K dalton cellular SV40 tumor antigen present in SV40-transformed cells and uninfected embryonal carcinoma cells. Cell 1979; 17: 43-52.
Reference Link - Melero JA, Stitt DT, Mangel WF, Carroll RB. Identification of new polypeptide species (48-55K) immunoprecipitable by antiserum to purified large T antigen and present in SV40-infected and -transformed cells. Virology 1979; 93: 466-480.
Reference Link - Rotter V, Witte ON, Coffman R, Baltimore D. Abelson murine leukemia virus-induced tumors elicit antibodies against a host cell protein, P50. J Virol 1980; 36: 547-555.
- Soussi T. p53 Antibodies in the sera of patients with various types of cancer: a review. Cancer Res 2000; 60: 1777-1788.
- Genini D, Sheeter D, Rought S, Zaunders JJ, Susin SA, Kroemer G et al. HIV induces lymphocyte apoptosis by a p53-initiated, mitochondrial-mediated mechanism. FASEB J 2001; 15: 5-6.
- Sato Y, Shirata N, Murata T, Nakasu S, Kudoh A, Iwahori S et al. Transient increases in p53-responsible gene expression at early stages of Epstein-Barr virus productive replication. Cell Cycle 2010; 9: 807-814.
Reference Link - Turpin E, Luke K, Jones J, Tumpey T, Konan K, Schultz-Cherry S. Influenza virus infection increases p53 activity: role of p53 in cell death and viral replication. J Virol 2005; 79: 8802-8811.
Reference Link - Lazo PA, Santos CR. Interference with p53 functions in human viral infections, a target for novel antiviral strategies? Rev Med Virol 2011; 21: 285-300.
- Sato Y, Tsurumi T. Genome guardian p53 and viral infections. Rev Med Virol 2012; DOI: 10.1002/rmv.1738.
Reference Link - Soria C, Estermann FE, Espantman KC, O'Shea CC. Heterochromatin silencing of p53 target genes by a small viral protein. Nature 2010; 466: 1076-1081.
Reference Link - Izumi T, Io K, Matsui M, Shirakawa K, Shinohara M, Nagai Y et al. HIV-1 viral infectivity factor interacts with TP53 to induce G2 cell cycle arrest and positively regulate viral replication. Proc Natl Acad Sci U S A 2010; 107: 20798-20803.
Reference Link - Pampin M, Simonin Y, Blondel B, Percherancier Y, Chelbi-Alix MK. Cross talk between PML and p53 during poliovirus infection: implications for antiviral defense. J Virol 2006; 80: 8582-8592.
Reference Link - Townsend PA, Scarabelli TM, Davidson SM, Knight RA, Latchman DS, Stephanou A. STAT-1 interacts with p53 to enhance DNA damage-induced apoptosis. J Biol Chem 2004; 279: 5811-5820.
Reference Link - Zhang F, Sriram S. Identification and characterization of the interferon-beta-mediated p53 signal pathway in human peripheral blood mononuclear cells. Immunology 2009; 128: e905-918.
Reference Link - Munoz-Fontela C, Macip S, Martinez-Sobrido L, Brown L, Ashour J, Garcia-Sastre A et al. Transcriptional role of p53 in interferon-mediated antiviral immunity. J Exp Med 2008; 205: 1929-1938.
Reference Link - Mori T, Anazawa Y, Iiizumi M, Fukuda S, Nakamura Y, Arakawa H. Identification of the interferon regulatory factor 5 gene (IRF-5) as a direct target for p53. Oncogene 2002; 21: 2914-2918.
Reference Link - Taura M, Eguma A, Suico MA, Shuto T, Koga T, Komatsu K et al. p53 regulates Toll-like receptor 3 expression and function in human epithelial cell lines. Mol Cell Biol 2008; 28: 6557-6567.
Reference Link - Yoon CH, Lee ES, Lim DS, Bae YS. PKR, a p53 target gene, plays a crucial role in the tumor-suppressor function of p53. Proc Natl Acad Sci U S A 2009; 106: 7852-7857.
Reference Link - Cuddihy AR, Li S, Tam NW, Wong AH, Taya Y, Abraham N et al. Double-stranded-RNA-activated protein kinase PKR enhances transcriptional activation by tumor suppressor p53. Mol Cell Biol 1999; 19: 2475-2484.
- Hummer BT, Li XL, Hassel BA. Role for p53 in gene induction by double-stranded RNA. J Virol 2001; 75: 7774-7777.
Reference Link - Pitha-Rowe IF, Pitha PM. Viral defense, carcinogenesis and ISG15: novel roles for an old ISG. Cytokine Growth Factor Rev 2007; 18: 409-417.
Reference Link - Obad S, Brunnstrom H, Vallon-Christersson J, Borg A, Drott K, Gullberg U. Staf50 is a novel p53 target gene conferring reduced clonogenic growth of leukemic U-937 cells. Oncogene 2004; 23: 4050-4059.
Reference Link - Petersson J, Lonnbro P, Herr AM, Morgelin M, Gullberg U, Drott K. The human IFN-inducible p53 target gene TRIM22 colocalizes with the centrosome independently of cell cycle phase. Exp Cell Res 2010; 316: 568-579.
Reference Link - Frank AK, Leu JI, Zhou Y, Devarajan K, Nedelko T, Klein-Szanto A et al. The codon 72 polymorphism of p53 regulates interaction with NF-kB and transactivation of genes involved in immunity and inflammation. Mol Cell Biol 2011; 31:1201-1213.
Reference Link - Donehower LA, Harvey M, Slagle BL, McArthur MJ, Montgomery CA, Jr., Butel JS et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 1992; 356: 215-221.
Reference Link - Munoz-Fontela C, Pazos M, Delgado I, Murk W, Mungamuri SK, Lee SW et al. p53 Serves as a Host Antiviral Factor That Enhances Innate and Adaptive Immune Responses to Influenza A Virus. Journal of Immunology 2011; 187: 6428-6436.
Reference Link - Tang X, Asano M, O'Reilly A, Farquhar A, Yang Y, Amar S. p53 is an important regulator of CCL2 gene expression. Curr Mol Med 2012; 12: 929-943.
Reference Link - Shiraishi K, Fukuda S, Mori T, Matsuda K, Yamaguchi T, Tanikawa C et al. Identification of fractalkine, a CX3C-type chemokine, as a direct target of p53. Cancer Research 2000; 60: 3722-3726.
- Mehta SA, Christopherson KW, Bhat-Nakshatri P, Goulet RJ, Jr., Broxmeyer HE, Kopelovich L et al. Negative regulation of chemokine receptor CXCR4 by tumor suppressor p53 in breast cancer cells: implications of p53 mutation or isoform expression on breast cancer cell invasion. Oncogene 2007; 26: 3329-3337.
Reference Link - Son DS, Kabir SM, Dong YL, Lee E, Adunyah SE. Inhibitory effect of tumor suppressor p53 on proinflammatory chemokine expression in ovarian cancer cells by reducing proteasomal degradation of IkappaB. PLoS One 2012; 7: e51116.
Reference Link - Son DS, Parl AK, Rice VM, Khabele D. Keratinocyte chemoattractant (KC)/human growth-regulated oncogene (GRO) chemokines and pro-inflammatory chemokine networks in mouse and human ovarian epithelial cancer cells. Cancer Biol Ther 2007; 6: 1302-1312.
Reference Link - Yeudall WA, Vaughan CA, Miyazaki H, Ramamoorthy M, Choi MY, Chapman CG et al. Gain-of-function mutant p53 upregulates CXC chemokines and enhances cell migration. Carcinogenesis 2012; 33: 442-451.
Reference Link - Mitchell RA, Liao H, Chesney J, Fingerle-Rowson G, Baugh J, David J et al. Macrophage migration inhibitory factor (MIF) sustains macrophage proinflammatory function by inhibiting p53: Regulatory role in the innate immune response. Proc Natl Acad Sci U S A 2002; 99: 345-350.
Reference Link - Zhang X, Liu X, Shang H, Xu Y, Qian M. Monocyte chemoattractant protein-1 induces endothelial cell apoptosis in vitro through a p53-dependent mitochondrial pathway. Acta Biochim Biophys Sin (Shanghai) 2011; 43: 787-795.
Reference Link - Acosta JC, O'Loghlen A, Banito A, Raguz S, Gil J. Control of senescence by CXCR2 and its ligands. Cell Cycle 2008; 7: 2956-2959.
Reference Link - Manes S, Mira E, Colomer R, Montero S, Real LM, Gomez-Mouton C et al. CCR5 expression influences the progression of human breast cancer in a p53-dependent manner. J Exp Med 2003; 198: 1381-1389.
Reference Link - Gorgoulis VG, Zacharatos P, Kotsinas A, Kletsas D, Mariatos G, Zoumpourlis V et al. p53 activates ICAM-1 (CD54) expression in an NF-kappa B-independent manner. EMBO Journal 2003; 22: 1567-1578.
Reference Link - Yakes FM, Wamil BD, Sun F, Yan HP, Carter CE, Hellerqvist CG. CM101 treatment overrides tumor-induced immunoprivilege leading to apoptosis. Cancer Res 2000; 60: 5740-5746.
- Rosenblum MD, Olasz E, Woodliff JE, Johnson BD, Konkol MC, Gerber KA et al. CD200 is a novel p53-target gene involved in apoptosis-associated immune tolerance. Blood 2004; 103: 2691-2698.
Reference Link - Donev RM, Cole DS, Sivasankar B, Hughes TR, Morgan BP. p53 regulates cellular resistance to complement lysis through enhanced expression of CD59. Cancer Res 2006; 66: 2451-2458.
Reference Link - Kadaja-Saarepuu L, Looke M, Balikova A, Maimets T. Tumor suppressor p53 down-regulates expression of human leukocyte marker CD43 in non-hematopoietic tumor cells. Int J Oncol 2012; 40: 567-576.
- Kadaja L, Laos S, Maimets T. Overexpression of leukocyte marker CD43 causes activation of the tumor suppressor proteins p53 and ARF. Oncogene 2004; 23: 2523-2530.
Reference Link - Menendez D, Shatz M, Azzam K, Garantziotis S, Fessler MB, Resnick MA. The Toll-Like Receptor Gene Family Is Integrated into Human DNA Damage and p53 Networks. PLoS Genet 2011; 7: e1001360.
Reference Link - Jordan JJ, Menendez D, Inga A, Nourredine M, Bell D, Resnick MA. Noncanonical DNA motifs as transactivation targets by wild type and mutant p53. PLoS Genet 2008; 4: e1000104.
Reference Link - Menendez D, Inga A, Resnick MA. The expanding universe of p53 targets. Nat Rev Cancer 2009; 9: 724-737.
Reference Link - Menendez D, Inga A, Snipe J, Krysiak O, Schonfelder G, Resnick MA. A single-nucleotide polymorphism in a half-binding site creates p53 and estrogen receptor control of vascular endothelial growth factor receptor 1. Mol Cell Biol 2007; 27: 2590-2600.
Reference Link - Menendez D, Krysiak O, Inga A, Krysiak B, Resnick MA, Schonfelder G. A SNP in the flt-1 promoter integrates the VEGF system into the p53 transcriptional network. Proc Natl Acad Sci USA 2006; 103: 1406-1411.
Reference Link - Shatz M, Menendez D, Resnick MA. The human TLR innate immune gene family is differentially influenced by DNA stress and p53 status in cancer cells. Cancer Res 2012; 72: 3948-3957.
Reference Link - Menendez D, Shatz M, Resnick MA. Interactions between the tumor suppressor p53 and immune responses. Current Opinion in Oncology 2013; 25: 85-92.
Reference Link - Ishizaki H, Song GY, Srivastava T, Carroll KD, Shahabi V, Manuel ER et al. Heterologous prime/boost immunization with p53-based vaccines combined with toll-like receptor stimulation enhances tumor regression. J Immunother 2010; 33: 609-617.
Reference Link - Zhu K, Wang J, Zhu J, Jiang J, Shou J, Chen X. p53 induces TAP1 and enhances the transport of MHC class I peptides. Oncogene 1999; 18: 7740-7747.
Reference Link - Iwaki S, Lu Y, Xie Z, Druey KM. p53 negatively regulates RGS13 protein expression in immune cells. J Biol Chem 2011; 286: 22219-22226.
Reference Link - Heinemann A, Zhao F, Pechlivanis S, Eberle J, Steinle A, Diederichs S et al. Tumor suppressive microRNAs miR-34a/c control cancer cell expression of ULBP2, a stress-induced ligand of the natural killer cell receptor NKG2D. Cancer Res 2012; 72: 460-471.
Reference Link - Textor S, Fiegler N, Arnold A, Porgador A, Hofmann TG, Cerwenka A. Human NK cells are alerted to induction of p53 in cancer cells by upregulation of the NKG2D ligands ULBP1 and ULBP2. Cancer Res 2011; 71: 5998-6009.
Reference Link - Li H, Lakshmikanth T, Garofalo C, Enge M, Spinnler C, Anichini A et al. Pharmacological activation of p53 triggers anticancer innate immune response through induction of ULBP2. Cell Cycle 2011; 10: 3346-3358.
Reference Link - Schetter AJ, Heegaard NH, Harris CC. Inflammation and cancer: interweaving microRNA, free radical, cytokine and p53 pathways. Carcinogenesis 2010; 31: 37-49.
Reference Link - Lu LF, Liston A. MicroRNA in the immune system, microRNA as an immune system. Immunology 2009; 127: 291-298.
Reference Link - Fabbri M, Bottoni A, Shimizu M, Spizzo R, Nicoloso MS, Rossi S et al. Association of a microRNA/TP53 feedback circuitry with pathogenesis and outcome of B-cell chronic lymphocytic leukemia. JAMA 2011; 305: 59-67.
Reference Link - Xu-Monette ZY, Medeiros LJ, Li Y, Orlowski RZ, Andreeff M, Bueso-Ramos CE et al. Dysfunction of the TP53 tumor suppressor gene in lymphoid malignancies. Blood 2012; 119: 3668-3683.
Reference Link - Caron de Fromentel C, May-Levin F, Mouriesse H, Lemerle J, Chandrasekaran K, May P. Presence of circulating antibodies against cellular protein p53 in a notable proportion of children with B-cell lymphoma. Int J Cancer 1987; 39: 185-189.
Reference Link - Crawford LV, Pim DC, Bulbrook RD. Detection of antibodies against the cellular protein p53 in sera from patients with breast cancer. Int J Cancer 1982; 30: 403-408.
Reference Link - Hollstein M, Sidransky D, Vogelstein B, Harris CC. p53 mutations in human cancers. Science 1991; 253: 49-53.
Reference Link - Ito D, Visus C, Hoffmann TK, Balz V, Bier H, Appella E et al. Immunological characterization of missense mutations occurring within cytotoxic T cell-defined p53 epitopes in HLA-A*0201+ squamous cell carcinomas of the head and neck. Int J Cancer 2007; 120: 2618-2624.
Reference Link - Dowell SP, Wilson PO, Derias NW, Lane DP, Hall PA. Clinical utility of the immunocytochemical detection of p53 protein in cytological specimens. Cancer Res 1994; 54: 2914-2918.
- Zambetti GP, Levine AJ. A comparison of the biological activities of wild-type and mutant p53. FASEB J 1993; 7: 855-865.
- Carbone DP, Ishida T, Chada S, Stipanov M, Gabrilovich DI. Dendritic cells transduced with wild type p53 gene elicit potent antitumor immune responses. Journal of Leukocyte Biology 1998: 11-11.
- Lauwen MM, Zwaveling S, de Quartel L, Ferreira Mota SC, Grashorn JA, Melief CJ et al. Self-tolerance does not restrict the CD4+ T-helper response against the p53 tumor antigen. Cancer Res 2008; 68: 893-900.
Reference Link - Cheok CF, Verma CS, Baselga J, Lane DP. Translating p53 into the clinic. Nat Rev Clin Oncol 2011; 8: 25-37.
Reference Link - Chiappori AA, Soliman H, Janssen WE, Antonia SJ, Gabrilovich DI. INGN-225: a dendritic cell-based p53 vaccine (Ad.p53-DC) in small cell lung cancer: observed association between immune response and enhanced chemotherapy effect. Expert Opin Biol Ther 2010; 10: 983-991.
Reference Link - Met O, Balslev E, Flyger H, Svane IM. High immunogenic potential of p53 mRNA-transfected dendritic cells in patients with primary breast cancer. Breast Cancer Res Treat 2011; 125: 395-406.
Reference Link - Nijman HW, Lambeck A, van der Burg SH, van der Zee AG, Daemen T. Immunologic aspect of ovarian cancer and p53 as tumor antigen. J Transl Med 2005; 3: 34.
Reference Link - Speetjens FM, Kuppen PJ, Welters MJ, Essahsah F, Voet van den Brink AM, Lantrua MG et al. Induction of p53-specific immunity by a p53 synthetic long peptide vaccine in patients treated for metastatic colorectal cancer. Clin Cancer Res 2009; 15: 1086-1095.
Reference Link - 116. Lane DP, Cheok CF, Lain S. p53-based cancer therapy. Cold Spring Harb Perspect Biol 2010; 2: a001222.
Reference Link - Vermeij R, Leffers N, van der Burg SH, Melief CJ, Daemen T, Nijman HW. Immunological and clinical effects of vaccines targeting p53-overexpressing malignancies. J Biomed Biotechnol 2011; 2011: 702146.
- Ak P, Levine AJ. p53 and NF-kappaB: different strategies for responding to stress lead to a functional antagonism. FASEB J 2010; 24: 3643-3652.
Reference Link - Gudkov AV, Gurova KV, Komarova EA. Inflammation and p53: A Tale of Two Stresses. Genes Cancer 2011; 2: 503-516.
Reference Link - Suzuki K, Murphy SH, Xia Y, Yokota M, Nakagomi D, Liu F et al. Tumor suppressor p53 functions as a negative regulator in IgE-mediated mast cell activation. PLoS One 2011; 6: e25412.
Reference Link - Murphy SH, Suzuki K, Downes M, Welch GL, De Jesus P, Miraglia LJ et al. Tumor suppressor protein (p)53, is a regulator of NF-kappaB repression by the glucocorticoid receptor. Proc Natl Acad Sci U S A 2011; 108: 17117-17122.
Reference Link - Madenspacher JH, Azzam KM, Gowdy KM, Malcolm CK, Nick JA, Dixon D et al. p53 integrates host defense and cell fate during bacterial pneumonia. J Experimental Medicine 2013; 210: 891-904.
Reference Link - Pan D, Pan LZ, Hill R, Marcato P, Shmulevitz M, Vassilev LT et al. Stabilisation of p53 enhances reovirus-induced apoptosis and virus spread through p53-dependent NF-kappaB activation. Br J Cancer 2011; 105: 1012-1022.
Reference Link - Wei J, Nagy TA, Vilgelm A, Zaika E, Ogden SR, Romero-Gallo J et al. Regulation of p53 tumor suppressor by Helicobacter pylori in gastric epithelial cells. Gastroenterology 2010; 139: 1333-1343.
Reference Link - Wei J, Noto J, Zaika E, Romero-Gallo J, Correa P, El-Rifai W et al. Pathogenic bacterium Helicobacter pylori alters the expression profile of p53 protein isoforms and p53 response to cellular stresses. Proc Natl Acad Sci U S A 2012; 109: E2543-2550.
Reference Link - Salminen A, Kauppinen A, Kaarniranta K. Emerging role of NF-kappaB signaling in the induction of senescence-associated secretory phenotype (SASP). Cell Signal 2012; 24: 835-845.
Reference Link - Kuilman T, Michaloglou C, Mooi WJ, Peeper DS. The essence of senescence. Genes Dev 2010; 24: 2463-2479.
Reference Link - Groppo R, Richter JD. CPEB Control of NF{kappa}B Nuclear Localization and IL-6 Production Mediates Cellular Senescence. Mol Cell Biol 2011; 31: 2707-2714
Reference Link - Kuilman T, Michaloglou C, Vredeveld LC, Douma S, van Doorn R, Desmet CJ et al. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 2008; 133: 1019-1031.
Reference Link - Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell 2011; 144: 646-674.
Reference Link - Hanahan D, Weinberg RA. The hallmarks of cancer. Cell 2000; 100: 57-70.
Reference Link - Hainaut P. Chapter 1: TP53 Coordinator of the Processes that Underlie the Hallmarks of Cancer. Hainaut P, Olivier M, Wiman KG (eds), p53 in the Clinics 2013; DOI 10.1007/978-1-4614-3676-8_1, Springer Science, New York, NY, USA: 1-23.
- Nikulenkov F, Spinnler C, Li H, Tonelli C, Shi Y, Turunen M et al. Insights into p53 transcriptional function via genome-wide chromatin occupancy and gene expression analysis. Cell Death Differ 2012; 19: 1992-2002.
Reference Link - Smeenk L, van Heeringen SJ, Koeppel M, Gilbert B, Janssen-Megens E, Stunnenberg HG et al. Role of p53 serine 46 in p53 target gene regulation. PLoS One 2011; 6: e17574.
Reference Link - Wei CL, Wu Q, Vega VB, Chiu KP, Ng P, Zhang T et al. A global map of p53 transcription-factor binding sites in the human genome. Cell 2006; 124: 207-219.
Reference Link - Jegga AG, Inga A, Menendez D, Aronow BJ, Resnick MA. Functional evolution of the p53 regulatory network through its target response elements. Proc Natl Acad Sci U S A 2008; 105: 944-949.
Reference Link - Lilyestrom W, Klein MG, Zhang R, Joachimiak A, Chen XS. Crystal structure of SV40 large T-antigen bound to p53: interplay between a viral oncoprotein and a cellular tumor suppressor. Genes Dev 2006; 20: 2373-2382.
Reference Link - Steegenga WT, Riteco N, Jochemsen AG, Fallaux FJ, Bos JL. The large E1B protein together with the E4orf6 protein target p53 for active degradation in adenovirus infected cells. Oncogene 1998; 16: 349-357.
Reference Link - Yi F, Saha A, Murakami M, Kumar P, Knight JS, Cai Q et al. Epstein-Barr virus nuclear antigen 3C targets p53 and modulates its transcriptional and apoptotic activities. Virology 2009; 388: 236-247.
Reference Link - Zhang Q, Gutsch D, Kenney S. Functional and physical interaction between p53 and BZLF1: implications for Epstein-Barr virus latency. Mol Cell Biol 1994; 14: 1929-1938.
- Scheffner M, Huibregtse JM, Vierstra RD, Howley PM. The HPV-16 E6 and E6-AP complex functions as a ubiquitin-protein ligase in the ubiquitination of p53. Cell 1993; 75: 495-505.
Reference Link - Friborg J, Jr., Kong W, Hottiger MO, Nabel GJ. p53 inhibition by the LANA protein of KSHV protects against cell death. Nature 1999; 402: 889-894.
- Takada S, Kaneniwa N, Tsuchida N, Koike K. Cytoplasmic retention of the p53 tumor suppressor gene product is observed in the hepatitis B virus X gene-transfected cells. Oncogene 1997; 15: 1895-1901.
Reference Link - McGivern DR, Lemon SM. Tumor suppressors, chromosomal instability, and hepatitis C virus-associated liver cancer. Annu Rev Pathol 2009; 4: 399-415.
Reference Link - Deng L, Nagano-Fujii M, Tanaka M, Nomura-Takigawa Y, Ikeda M, Kato N et al. NS3 protein of Hepatitis C virus associates with the tumour suppressor p53 and inhibits its function in an NS3 sequence-dependent manner. J Gen Virol 2006; 87: 1703-1713.
Reference Link - Ariumi Y, Kaida A, Hatanaka M, Shimotohno K. Functional cross-talk of HIV-1 Tat with p53 through its C-terminal domain. Biochem Biophys Res Commun 2001; 287: 556-561.
Reference Link - Wang X, Shen Y, Qiu Y, Shi Z, Shao D, Chen P et al. The non-structural (NS1) protein of influenza A virus associates with p53 and inhibits p53-mediated transcriptional activity and apoptosis. Biochem Biophys Res Commun 2010; 395: 141-145.
Reference Link - Pise-Masison CA, Choi KS, Radonovich M, Dittmer J, Kim SJ, Brady JN. Inhibition of p53 transactivation function by the human T-cell lymphotropic virus type 1 Tax protein. J Virol 1998; 72: 1165-1170.
- Sun M, Fuentes SM, Timani K, Sun D, Murphy C, Lin Y et al. Akt plays a critical role in replication of nonsegmented negative-stranded RNA viruses. J Virol 2008; 82: 105-114.
Reference Link