Introduction
Progesterone is released from cumulus cells surrounding the oocyte and has been demonstrated to act as a chemoattractant for human and rabbit sperm [
Studies examining the effects of progesterone on boar sperm cells have largely focussed on its effects on the acrosome reaction [
Material and Methods
Semen supply
Semen was supplied commercially by The Pig Improvement Company (PIC) from their stock of boars (PIC337 Champion species). Semen was diluted 1:1 upon collection into TriXCell (IMV International, Minneapolis, MN, USA) semen extender according to the manufacturer instructions. Semen was delivered overnight in flatpacks at ambient temperature. Upon receipt, the diluted semen was then aliquoted into 15ml polypropylene centrifuge tubes and stored at 16°C in accordance with the manufacturer instructions until use. Assays were completed within 2 days of delivery. No information is available regarding their fertility.
Vitality test
Vitality testing using propridium iodide (PI) staining based on the method by Pintado and colleagues [
Acrosome staining
The acrosome status of sperm was investigated using the method of He et al [
Mobility assay solutions
The mobility assay was based on the validated method of Vizcarra & Ford [
Mobility assay
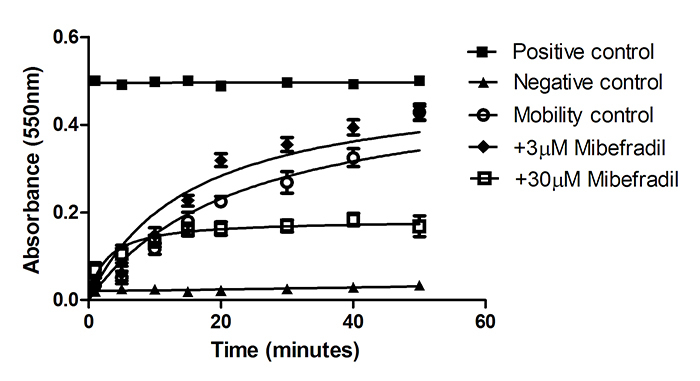
Cuvettes containing the working Accudenz solution were pre-warmed to 37°C. Data presented in each figure were derived using a split-ejaculate method such that cells under control and treatment conditions were taken from the same insemination dose. 1×108 live sperm cells (in maximally 150µl) were carefully pipetted onto the surface of the Accudenz solution and the change in absorbance (550nm) over time was monitored using a spectrophotometer. This treatment is hereafter called the mobility control. A negative control was prepared by first heating 1×108 sperm cells at 60°C for 60 minutes. A positive control was prepared by mixing 1×108 cells with 1.5ml working Accudenz solution prior to loading into a cuvette. Progesterone was added to the working Accudenz solution to a final concentration of 1, 10 or 100nM and used to monitor sperm penetration rate in response to progesterone concentration. Absorbance readings were recorded at time intervals over 50 minutes (1, 5, 10, 15, 20, 30, 40 and 50 minutes) and cuvettes were incubated at 37°C for the duration of this time period.
To construct data sets within a figure, multiple runs were taken from an individual boar sample and multiple boars were used as indicated in the figure legends. A total of 17 different boars were used by the end of the study.
Capacitating conditions
Capacitating media (CM) was composed of 5mM KCl, 1mM KH2PO4, 95mM NaCl, 5.55mM glucose, 25mM NaHCO3, 2mM CaCl2, 0.4% BSA and 2.5mM pyruvate (pH 7.4). Non-capacitating media (NCM) lacked calcium, bicarbonate and BSA and consisted of 2.7mM KCl, 1.5mM KH2PO4, 8.1mM Na2HPO4, 137mM NaCl, 5.55mM glucose and 2.5mM pyruvate (pH 7.4). 1×108 cells were pelleted and resuspended in 1ml of either CM or NCM and incubated at 37°C for 3hours.
Following capacitation the samples were pelleted, resuspended in mobility buffer and the mobility assay ran as described for non-capacitated cells.
SDS-PAGE and Western Blotting
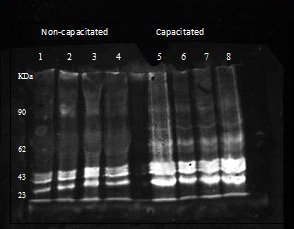
1×108 cells were incubated in NCM or CM for 3 hours, pelleted at 17000 × g then heated at 95°C in Laemmli sample buffer and frozen for subsequent use in SDS-PAGE. Sperm proteins were separated on 10% polyacrylamide gels and transferred to PVDF membranes by electrophoresis. The membranes were incubated overnight at 4°C with anti phospho-tyrosine primary antibodies (Cell Signaling Technology, UK) at 1:20 000 dilution in Tris-buffered saline (25mM Tris-HCl pH7.4, 150mM NaCl) containing 0.1%(v/v) Tween 20 and 5%(w/v) bovine serum albumin. The detection method involved the use of a WesternDot™625 western blot kit in accordance with the manufacturer instructions (Life Technologies, UK) and imaging with a Biorad Gel Doc XR system.
Data analysis
Data represented mean ± SEM for four ejaculates measured in quadruplet (Graphpad Prism). Curves are fitted using non-linear regression model and Km values (minutes) represent the time at which absorbance was half maximal (Graphpad Prism). P < 0.05 is taken as statistically significant.
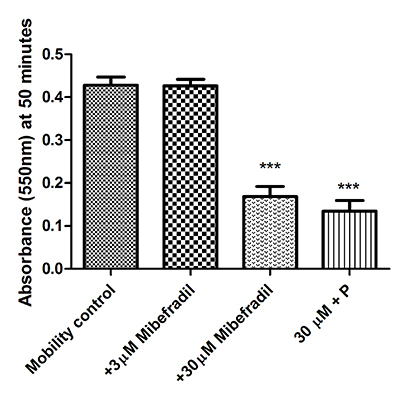
To test for differences in the rate of mobility between control and progesterone treated sperm cells data were first transformed to linearize then an ANCOVA was carried out on regression plots of the linearized data using R (open source statistical software http://www.r-project.org/. R version 2.15.1 (2012-06-22) “Roasted Marshmallows”. Copyright 2012 The R Foundation for Statistical Computing). Data were linearized using ln(y) = m*ln(x)+ln(c), where y = mean absorbance and x = 1/time (min). The effect of mibefradil on sperm mobility was analysed using ANOVA followed by Tukeys post-hoc test for three time points (10, 20 and 50 minutes).
Chemicals and reagents
Unless stated all chemicals and reagents were obtained from Sigma-Aldrich, UK.
Mibefradil was obtained from Tocris Bioscience, Bristol, UK.
Results
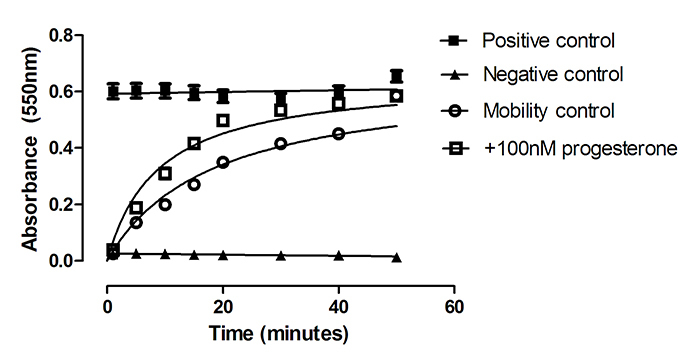
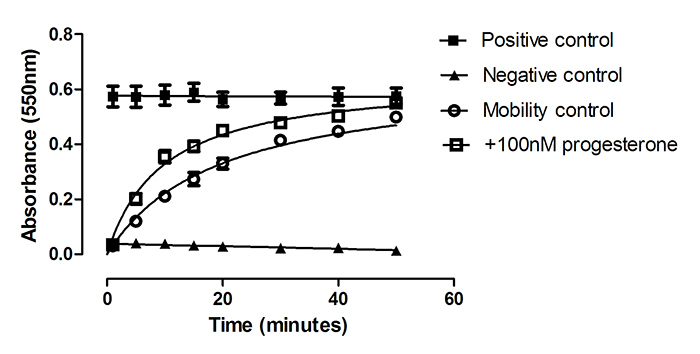
Non-capacitated boar sperm penetrated the working Accudenz solution with a Km = 18.0±2.2 min. Accudenz solutions containing increasing concentrations of progesterone (1, 10, 100nM) were prepared in order to monitor a change in mobility induced by this putative chemoattractant. Only 100nM progesterone treatment produced a significant increase in sperm mobility as measured by an increase in absorption over time in the sperm mobility assay (Figure 1). Analysis of the progesterone effect using ANCOVA required linearised plots so data from Figure 1 was first transformed (ln(y) = m*ln(x)+ln(c) where y = mean absorbance and x = 1/time (min)) and the gradient and intercept of the regression lines were then compared. The independent variable in the regression analysis was ln(1/time), the dependent variable was ln(mean absorbance) and the factor was +/-progesterone treatment. Initial ANCOVA analysis revealed that there was no significant interaction between progesterone treatment and time (p = 0.76) and therefore progesterone treatment could be analysed for its effect alone on mobility. For non-capacitated sperm, the intercepts of the regression lines were significantly higher in the presence of 100nM progesterone. The intercept of the regression line for hormone treatment was -3.32 and for controls, the intercept was -3.49, (F= 8.997, p= 0.01). This result indicates progesterone significantly increases the rate of penetration of non-capacitated cells. Indeed 100nM progesterone caused over a two-fold increase in the rate of penetration rate (Km = 8.0±0.8min). There were no significant differences between the gradients for 100nM progesterone treated and control sperm for either capacitated (see below) or non-capacitated sperm indicating that there was no interaction between hormone treatment and time and that the relationship between absorbance and time could be modelled by a simple additive regression model without variable interaction.
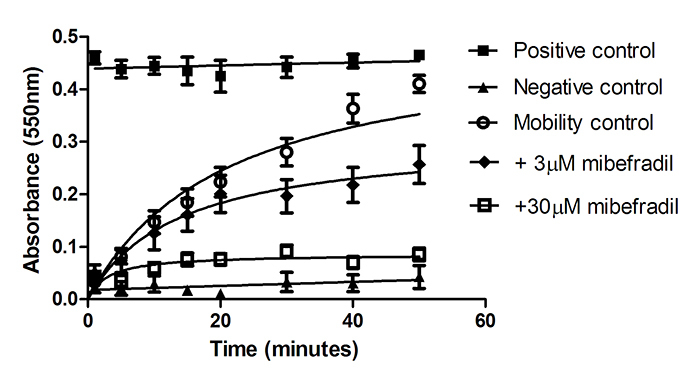
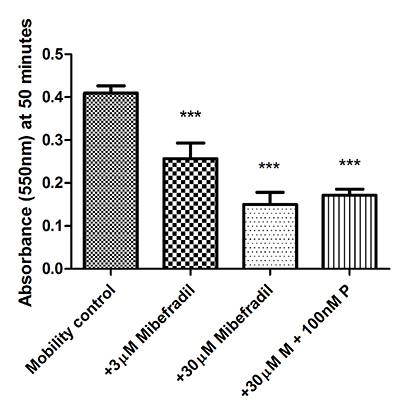
CatSper is sensitive to the Cav blocker mibefradil [
Capacitation is associated with alterations of the membrane of sperm [
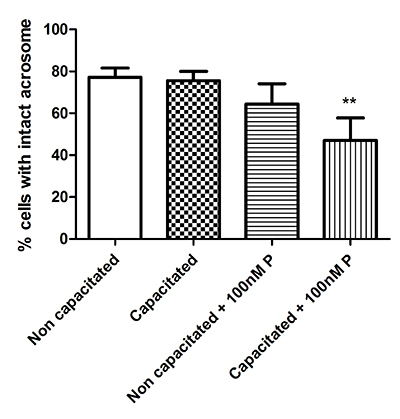
Progesterone (100nM) induced the acrosome reaction in significantly greater number of cells in capacitated cells only, although approximately half of the population remained unreacted (Figure 7). Finally, the capacitation status of the cells was confirmed by assessing the total protein tyrosine phosphorylation status (Figure 8).
Discussion
Progesterone has been demonstrated to induce a number of calcium dependent mechanisms in sperm including hyperactivation [
Previous studies examining the effect of progesterone on human sperm cell motility have typically involved the use of CASA systems and report significant effects on hyperactivation [
As prematurely acrosome reacted sperm cannot undertake fertilisation [
The pattern of response to progesterone of boar sperm cells is interesting due to the molecular mechanism by which progesterone has been proposed to act in stimulating sperm motility. Progesterone potentiates CatSper-mediated currents in human but not mouse sperm, with the constitutive activity of CatSper being much higher in the latter species. CatSper is conserved between species [
In summary, for the first time we have demonstrated that progesterone can sustainably increase the mobility of boar sperm cells in an extracellular calcium dependent manner and that the increased distance travelled with time presents a selective advantage for cells that remain acrosome intact.
Acknowledgements
The authors would like to thank The Carnegie Trust for the Universities of Scotland for the studentship award for JMC which permitted her to complete her work.
References
- Teves ME, Barbano F, Guidobaldi HA, Sanchez R, Miska W, Giojalas LC. Progesterone at the picomolar range is a chemoattractant for mammalian spermatozoa. Fertil Steril 2006; 86: 745-749.
Reference Link - Guidobaldi HA, Teves ME, Unates DR, Anastasia A, Giojalas LC. Progesterone from the cumulus cells is the sperm chemoattractant secreted by the rabbit oocyte cumulus complex. PLoS One 2008; 3: e3040.
Reference Link - Oren-Benaroya R, Orvieto R, Gakamsky A, Pinchasov M, Eisenbach M. The sperm chemoattractant secreted from human cumulus cells is progesterone. Hum Reprod 2008; 23: 2339-2345.
Reference Link - Gakamsky A, Armon L, Eisenbach M. Behavioral response of human spermatozoa to a concentration jump of chemoattractants or intracellular cyclic nucleotides. Hum Reprod 2009; 24: 1152-1163.
Reference Link - Cohen-Dayag A, Ralt D, Tur-Kaspa I, Manor M, Makler A, Dor J, Mashiach S, Eisenbach M. Sequential acquisition of chemotactic responsiveness by human spermatozoa. Biol Reprod 1994; 50: 786-790.
Reference Link - Cohen-Dayag A, Tur-Kaspa I, Dor J, Mashiach S, Eisenbach M. Sperm capacitation in humans is transient and correlates with chemotactic responsiveness to follicular factors. Proc Natl Acad Sci U S A 1995; 92: 11039-11043.
Reference Link - Uhler ML, Leung A, Chan SY, Wang C. Direct effects of progesterone and antiprogesterone on human sperm hyperactivated motility and acrosome reaction. Fertil Steril 1992; 58: 1191-1198.
- Armon L, Eisenbach M. Behavioral mechanism during human sperm chemotaxis: involvement of hyperactivation. PLoS One 2011; 6: e28359.
Reference Link - Harper CV, Barratt CL, Publicover SJ. Stimulation of human spermatozoa with progesterone gradients to simulate approach to the oocyte. Induction of [Ca(2+)](i) oscillations and cyclical transitions in flagellar beating. J Biol Chem 2004; 279: 46315-46325.
Reference Link - Publicover S, Harper CV, Barratt C. [Ca2+]i signalling in sperm--making the most of what you've got. Nat Cell Biol 2007; 9: 235-242.
Reference Link - Lishko PV, Botchkina IL, Kirichok Y. Progesterone activates the principal Ca2+ channel of human sperm. Nature 2011; 471: 387-391.
Reference Link - Strunker T, Goodwin N, Brenker C, Kashikar ND, Weyand I, Seifert R et al. The CatSper channel mediates progesterone-induced Ca2+ influx in human sperm. Nature 2011; 471: 382-386.
Reference Link - Kobori H, Miyazaki S, Kuwabara Y. Characterization of intracellular Ca(2+) increase in response to progesterone and cyclic nucleotides in mouse spermatozoa. Biol Reprod 2000; 63: 113-120.
Reference Link - Kim JC, Li Y, Lee S, Yi YJ, Park CS, Woo SH. Effects of cryopreservation on Ca2+ signals induced by membrane depolarization, caffeine, thapsigargin and progesterone in boar spermatozoa. Mol Cells 2008; 26: 558-565.
- Noguchi T, Fujinoki M, Kitazawa M, Inaba N. Regulation of hyperactivation of hamster spermatozoa by progesterone. Reprod Med Biol 2008; 7: 63-74.
Reference Link - Cheng FP, Gadella BM, Voorhout WF, Fazeli A, Bevers MM, Colenbrander B. Progesterone-induced acrosome reaction in stallion spermatozoa is mediated by a plasma membrane progesterone receptor. Biol Reprod 1998; 59: 733-742.
Reference Link - Sirivaidyapong S, Bevers MM, Colenbrander B. Acrosome reaction in dog sperm is induced by a membrane-localized progesterone receptor. J Androl 1999; 20: 537-544.
- Patrat C, Serres C & Jouannet P. The acrosome reaction in human spermatozoa. Biol Cell 2000; 92: 255-266.
Reference Link - Therien I, Manjunath P. Effect of progesterone on bovine sperm capacitation and acrosome reaction. Biol Reprod 2003; 69: 1408-1415.
Reference Link - Pizzari T. Of mice and sperm. Proc Natl Acad Sci U S A 2006; 103: 14983-14984.
Reference Link - Jang S, Yi LS. RU486 suppresses progesterone-induced acrosome reaction in boar spermatozoa. J Biochem Mol Biol 2002; 35: 604-608.
Reference Link - Jang S, Yi LS. Identification of a 71 kDa protein as a putative non-genomic membrane progesterone receptor in boar spermatozoa. J Endocrinol 2005; 184: 417-425.
Reference Link - Wu JT, Chiang KC, Cheng FP. Expression of progesterone receptor(s) during capacitation and incidence of acrosome reaction induced by progesterone and zona proteins in boar spermatozoa. Anim Reprod Sci 2006; 93: 34-45.
Reference Link - Ramio L, Rivera MM, Ramirez A Concha II, Peña A, Rigau T et al. Dynamics of motile-sperm subpopulation structure in boar ejaculates subjected to "in vitro" capacitation and further "in vitro" acrosome reaction. Theriogenology 2008; 69: 501-512.
Reference Link - Mortimer ST. A critical review of the physiological importance and analysis of sperm movement in mammals. Hum Reprod Update 1997; 3: 403-439.
Reference Link - Vizcarra JA, Ford JJ. Validation of the sperm mobility assay in boars and stallions. Theriogenology 2006; 66: 1091-1097.
Reference Link - Pintado B, de la Fuente J, Roldan ER. Permeability of boar and bull spermatozoa to the nucleic acid stains propidium iodide or Hoechst 33258, or to eosin: accuracy in the assessment of cell viability. J Reprod Fertil 2000; 118: 145-152.
Reference Link - He L, Bailey JL, Buhr MM. Incorporating lipids into boar sperm decreases chilling sensitivity but not capacitation potential. Biol Reprod 2001; 64: 69-79.
Reference Link - Abou-haila A, Tulsiani DR. Signal transduction pathways that regulate sperm capacitation and the acrosome reaction. Arch Biochem Biophys 2009; 485: 72-81.
Reference Link - Fujinoki M. Suppression of progesterone-enhanced hyperactivation in hamster spermatozoa by estrogen. Reproduction 2010; 140: 453-464.
Reference Link - Tesarik J, Moos J, Mendoza C. Stimulation of protein tyrosine phosphorylation by a progesterone receptor on the cell surface of human sperm. Endocrinology 1993; 133: 328-335.
Reference Link - Bonaccorsi L, Krausz C, Pecchioli P, Forti G, Baldi E. Progesterone-stimulated intracellular calcium increase in human spermatozoa is protein kinase C-independent. Mol Hum Reprod 1998; 4: 259-268.
Reference Link - Moseley FL, Jha KN, Bjorndahl L, Brewis IA, Publicover SJ, Barratt CL et al. Protein tyrosine phosphorylation, hyperactivation and progesterone-induced acrosome reaction are enhanced in IVF media: an effect that is not associated with an increase in protein kinase A activation. Mol Hum Reprod 2005; 11: 523-529.
Reference Link - Teves ME, Guidobaldi HA, Unates DR, Sanchez R, Miska W, Publicover SJ et al. Molecular mechanism for human sperm chemotaxis mediated by progesterone. PLoS One 2009; 4: e8211.
Reference Link - Carlson AE, Westenbroek RE, Quill T, Ren D, Clapham DE, Hille B et al. CatSper1 required for evoked Ca2+ entry and control of flagellar function in sperm. Proc Natl Acad Sci U S A 2003 100: 14864-14868.
Reference Link - Carlson AE, Quill TA, Westenbroek RE, Schuh SM, Hille B, Babcock DF. Identical phenotypes of CatSper1 and CatSper2 null sperm. J Biol Chem 2005; 280: 32238-32244.
Reference Link - Qi H, Moran MM, Navarro B, Chong JA, Krapivinsky G, Krapivinsky L et al. All four CatSper ion channel proteins are required for male fertility and sperm cell hyperactivated motility. Proc Natl Acad Sci U S A 2007; 104(4): 1219-1223.
Reference Link - Xia J, Reigada D, Mitchell CH, Ren D. CATSPER channel-mediated Ca2+ entry into mouse sperm triggers a tail-to-head propagation. Biol Reprod 2007; 77: 551-559.
Reference Link - Xia J, Ren D. The BSA-induced Ca2+ influx during sperm capacitation is CATSPER channel-dependent. Reprod Biol Endocrinol 2009; : 119.
Reference Link - Xia J, Ren D. Egg coat proteins activate calcium entry into mouse sperm via CATSPER channels. Biol Reprod 2009; 80: 1092-1098.
Reference Link - Bertoldo M, Holyoake PK, Evans G, Grupen CG. Follicular progesterone levels decrease during the period of seasonal infertility in sows. Reprod Domest Anim 2011; 46: 489-494.
Reference Link - Brown J, Cebra-Thomas JA, Bleil JD, Wassarman PM, Silver LM. A premature acrosome reaction is programmed by mouse t haplotypes during sperm differentiation and could play a role in transmission ratio distortion. Development 1989; 106: 769-773.
- Cai X, Clapham DE. Evolutionary genomics reveals lineage-specific gene loss and rapid evolution of a sperm-specific ion channel complex: CatSpers and CatSperbeta. PLoS One 2008; 3: e3569.
Reference Link - Song C, Gao B, Wu H, Xie Y, Wang X, Li B et al. Molecular cloning, spatial and temporal expression analysis of CatSper genes in the Chinese Meishan pigs. Reprod Biol Endocrinol 2011; 9: 132.
Reference Link - Darszon A, Acevedo JJ, Galindo BE, Hernández-González EO, Nishigaki T, Treviño CL, et al. Sperm channel diversity and functional multiplicity. Reproduction 2006; 131: 977-988.
Reference Link - Bernabo N, Pistilli MG, Mattioli M, Barboni B. Role of TRPV1 channels in boar spermatozoa acquisition of fertilizing ability. Mol Cell Endocrinol 2010; 323: 224-231.
Reference Link